Introduction
Antibodies 2020, 9(3), 36; https://doi.org/10.3390/antib9030036 (registering DOI)
Received: 26 June 2020 / Revised: 21 July 2020 / Accepted: 23 July 2020 / Published: 29 July 2020
Authors
The paper is authored by Vaneet K. Sharma (1), Bijay Misra (2), Kevin T. McManus (3), Sreenivas Avula (1), Kaliappanadar Nellaiappan (2), Marina Caskey (4), Jill Horowitz (4), Michel C. Nussenzweig (4), Michael S. Seaman (3), Indu Javeri (2) and Antu K. Dey (1)
Authors’ Belonged Companies
1 IAVI, 125 Broad Street, New York, NY 10004, USA
2 CuriRx, Inc., 205 Lowell Street, Wilmington, MA 01887, USA
3 Center for Virology and Vaccine Research, Beth Israel Deaconess Medical Center, Boston, MA 02215, USA
4 Laboratory of Molecular Immunology, The Rockefeller University, New York, NY 10065, USA
5 Howard Hughes Medical Institute, The Rockefeller University, New York, NY 10065, USA
* Author to whom correspondence is addressed with corresponding company numbers behind their names.
Abstract
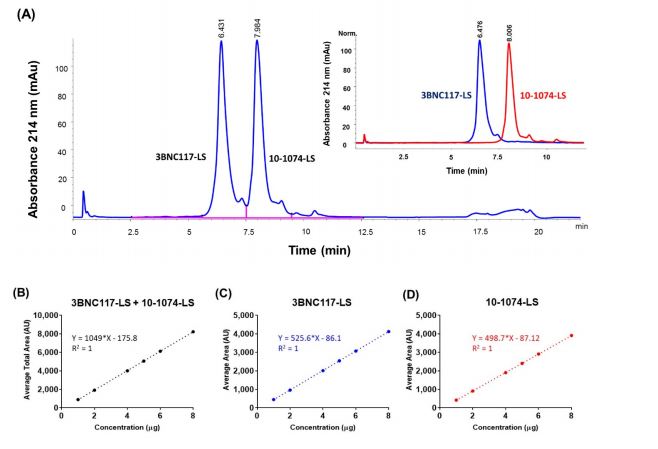
The discovery of numerous potent and broad neutralizing antibodies (bNAbs) against Human Immunodeficiency Virus type 1 (HIV-1) envelope glycoprotein has invigorated the potential of using them as an effective preventative and therapeutic agent. The majority of the anti-HIV-1 antibodies, currently under clinical investigation, are formulated singly for intravenous (IV) infusion. However, due to the high degree of genetic variability in the case of HIV-1, a single broad neutralizing antibody will likely not be sufficient to protect against the broad range of viral isolates. To that end, delivery of two or more co-formulated bnAbs against HIV-1 in a single subcutaneous (SC) injection is highly desired. We, therefore, co-formulated two anti-HIV bnAbs, 3BNC117-LS, and 10-1074-LS, to a total concentration of 150 mg/mL for SC administration and analyzed them using a panel of analytical techniques. Chromatographic-based methods, such as RP-HPLC, CEX-HPLC, and SEC-HPLC, were developed to ensure the separation and detection of each antibody in the co-formulated sample. In addition, we used a panel of diverse pseudoviruses to detect the functionality of individual antibodies in the co-formulation. We also used these methods to test the stability of the co-formulated antibodies and believe that such an approach can support future efforts toward the formulation and characterization of multiple high-concentration antibodies for SC delivery.
View Full-Text